1. Complex reaction mechanisms of electrolyte decomposition at large-scale Ni-rich Li-ion battery cells including electrode crosstalk effect
Surat Prempluem, Thitiphum Sangsanit, Kanruthai Santiyuk, Kan Homlamai, Worapol Tejangkura, Ronnachai Songthan, Nichakarn Anansuksawat, Montree Sawangphruk
Journal of Power Sources, 2024, 606, 234538
DOI: https://doi.org/10.1016/j.jpowsour.2024.234538
Abstract: Ni-rich cathodes are essential for the advancement of high-energy lithium-ion batteries but encounter significant challenges at elevated voltages, including oxygen liberation and enhanced surface reactivity, which precipitate electrolyte decomposition. This study explores the intricate reaction mechanisms of electrolyte decomposition linked to the pronounced release of oxygen from the cathode lattice in large-scale 18650 cylindrical cells employing Ni-rich LiNi0.90Mn0.05Co0.05O2(NMC90)//graphite configurations, focusing on electrode crosstalk effects at high voltages. Notably, acetals such as methoxy methanol (49.93 %) and methane diol (50.07 %) were identified at 4.7 V, with their presence escalating at 4.9 V. Formaldehyde, present at 7.21 %, along with methanol (22.77 %), methane diol (36.35 %), and methoxy methanol (33.67 %), begins to manifest at 4.9 V. The dissolution of transition metals was analyzed using Inductively Coupled Plasma Atomic Emission Spectrometry, and changes in the double-layer capacitance were assessed through electrochemical impedance spectroscopy, further validating the link between these decomposition products and cathode failure associated with oxygen release. These insights reveal the complex relationship between electrolyte decomposition and cathode degradation due to oxygen release, highlighting a critical failure mechanism in large-scale cylindrical lithium-ion batteries.

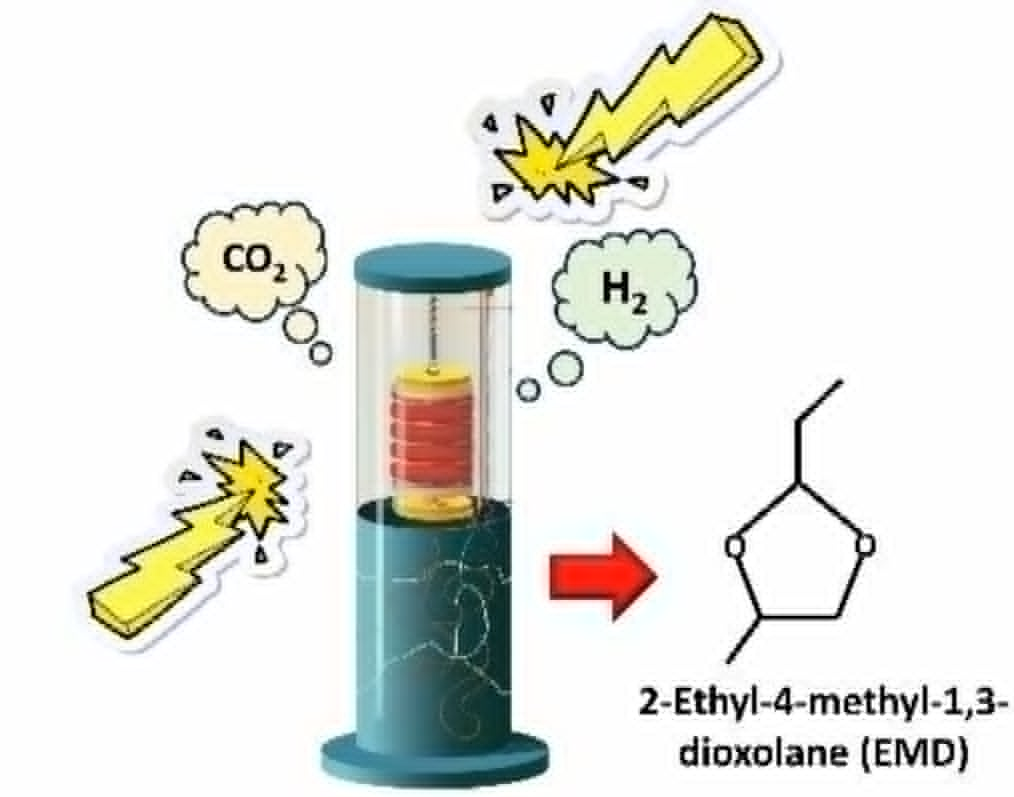
2. Unveiling a Novel Decomposition Pathway in Propylene Car-bonate-Based Supercapacitors: Insights from a Jelly Roll Config-uration Study
Phatsawit Wuamprakhona, Jiraporn Phojaroena, Thitiphum Sangsanita, Kanruthai Santiyuka, Kan Hom-lamaia, Worapol Tejangkuraa,and Montree Sawangphruka
ChemSusChem, 2024, N/N
DOI: https://doi.org/10.1002/cssc.202400053
Abstract: This research elucidates novel insights into the electrochemical properties and degradation phenomena of propylene carbonate (PC)-based supercapacitors at a large-scale 18650 cylindrical jelly-roll cell level. Central to our findings is the identification of 2-ethyl-4-methyl-1,3-dioxolane (EMD) as a hitherto undocumented decomposition by-product, highlighting the nuanced complexity of PC electrolyte stability. We further demonstrate that elevated operational voltages precipitate accelerated electrolyte degradation, underscoring the criticality of defining the operational voltage window for maximizing device longevity. Employing advanced analytical techniques, including gas chromatography-mass spectrometry (GC-MS), this study meticulously analyzes electrolyte decomposition mechanisms. The outcomes offer pivotal insights into the operational constraints and chemical resilience of PC-based supercapacitors, contributing significantly to the optimization of supercapacitor design and application. By delineating a specific decomposition pathway, this investigation enriches the understanding of electrochemical dynamics in supercapacitor systems, providing a foundation for future research and technological advancement in energy storage devices.
3. How Uniform Particle Size of NMC90 Boosts Li-ion Mobility for Faster Charging and Discharging at a Cylindrical Li-ion Battery Cell
Nichakarn Anansuksawat, Thitiphum Sangsanit, Surat Prempluem, Kan Homlamai, Worapol Tejangkura, and Montree Sawangphruk
Chemical Science, 2024, 2024,15, 2026-2036
DOI: https://doi.org/10.1039/D3SC05698H
Abstract: The pursuit of high-performing cathode materials for next-generation lithium ion batteries has focused on increasing the nickel content of the material. However, this study reveals that particle size uniformity is a more decisive factor in battery performance than the nickel content alone. Using in operando X-ray diffraction, in situ gas evolution, and Atlung intercalant diffusion, we compared two promising cathode materials: LiNi0.8Mn0.1Co0.1O2 (NMC80) and LiNi0.9Mn0.05Co0.05O2 (NMC90). We found that the NMC90 cell, with its more uniform particle size, exhibits a remarkable accumulation energy density of 558 kW h kgNMC−1, which is 22% higher than that of the NMC80 cell. Also, the NMC90 cell unexpectedly has a 20% better capacity retention, a 50-fold higher discharge capacity at the 5C rate, and an Atlung lithium diffusion coefficient that is one order of magnitude higher after 1000 cycles. In situ gas analysis at a high voltage of 4.5 V reveals that the NMC80 cell generates 1.75 times more CO2 than the NMC90 cell. These findings illuminate the intricate relationship between the nickel content, particle size uniformity, and battery performance. They offer vital insights for optimizing cathode materials in future lithium ion batteries.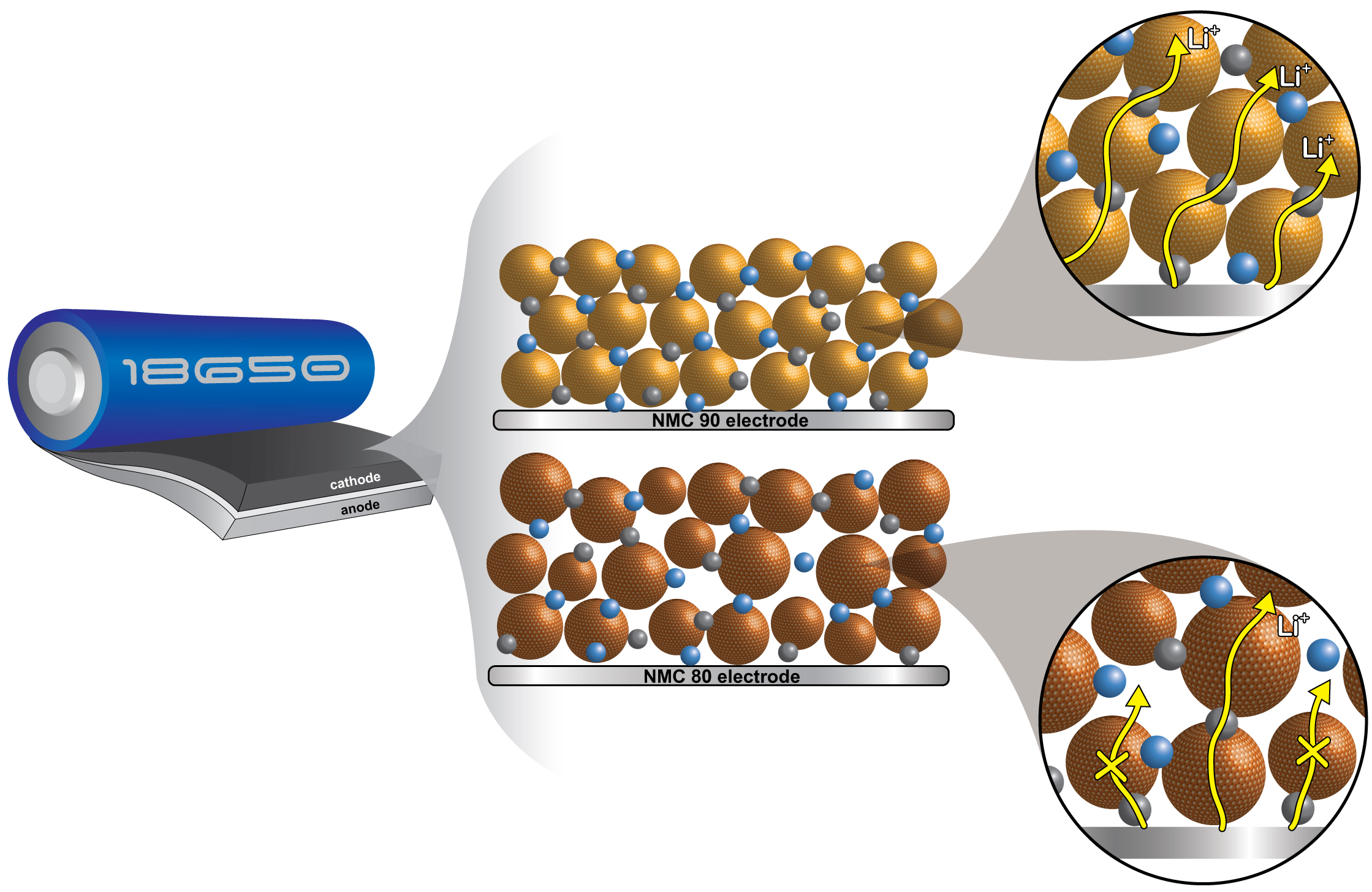

4. Non-flammable electrolyte for large-scale Ni-rich Li-ion batteries: Reducing thermal runaway risks
Thitiphum Sangsanit, Kan Homlamai, Nattanon Joraleechanchai, Surat Prempluem, Worapol Tejangkura, Montree Sawangphruk
Journal of Power Sources, 2024, 594, 234021
DOI: https://doi.org/10.1016/j.jpowsour.2023.234021
Abstract: We present a novel, non-flammable electrolyte for Ni-rich 18650 cylindrical lithium-ion batteries (LIBs) based on a blend of triethyl phosphate (TEP) and fluorinated ethylene carbonate (FEC). We conducted a comprehensive analysis of the electrolyte, including electrochemical tests, safety evaluations, electrode surface characterizations, and quantum calculations, to elucidate its property-function relationship. We found that combining TEP with a concentrated FEC as a co-solvent (1.2 M LiPF6 in TEP/FEC, 30/70 %v) resulted in a robust solid electrolyte interface (SEI) predominantly composed of lithium fluoride (LiF). This innovation improved the SEI's stability and suppressed electrolyte decomposition during extended cycling, leading to consistent battery performance. Our findings introduce a promising non-flammable electrolyte solution for LIBs that offers both thermal runaway prevention and seamless integration into existing battery manufacturing processes. This positions it as a potential alternative to solid-state electrolytes in advanced battery technology, with the potential to revolutionize the field.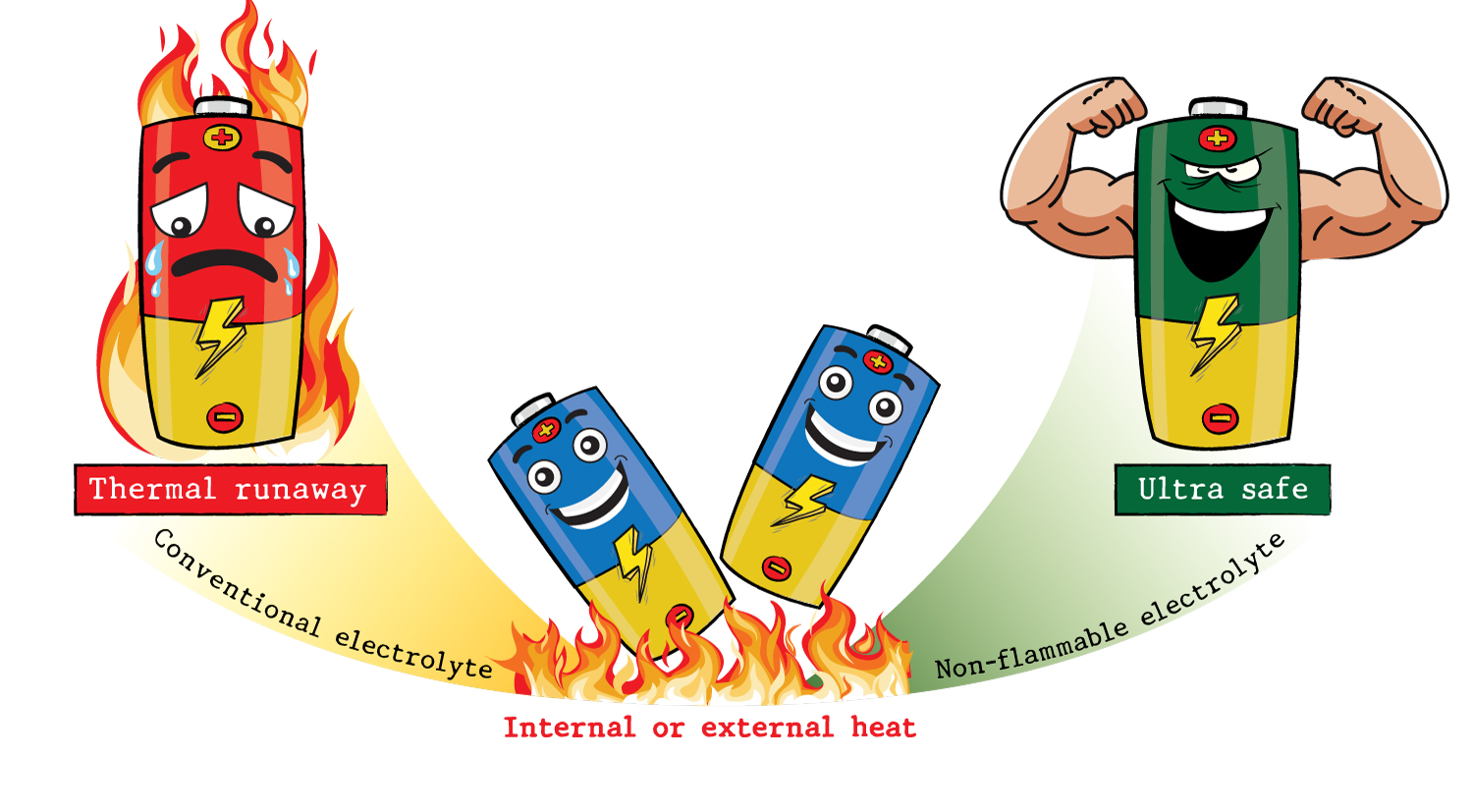

5. Rocking chair-type aqueous sodium-ion capacitors with biomass-derived activated carbon and Na3V2 (PO4) 2F3 nanoflower in a water-in-salt electrolyte
Arisa Phukhrongthung, Montree Sawangphruk, Pawin Iamprasertkun, Charuayporn Santhaweesuk, Channarong Puchongkawarin, Santamon Luanwuthi
Journal of Energy Storage, 2024, 80, 110369
DOI: https://doi.org/10.1016/j.est.2023.110369
Abstract: Sodium-ion capacitors (SICs) are promising energy storage devices that combine the characteristics of high-energy batteries and high-power capacitors, utilising abundant and cost-effective sodium resources. However, conventional SICs employ flammable and toxic organic electrolytes and an electrolyte-consuming mechanism. This work introduces a novel rocking chair-type aqueous sodium-ion capacitor (RC-ASIC) that employs a water-in-salt electrolyte (17 m NaClO4). It incorporates oil palm leaf-derived activated carbon (OPL_AC) as the anode and Na3(VPO4)2F3 with a flower-like structure (NVPF_NF) as the cathode. This RC-ASIC demonstrates a significant improvement in specific energy by extending the voltage window to 2.2 V. Furthermore, the cell with rocking-chair construction requires minimal electrolyte because sodium ions deintercalate from the cathode and adsorb onto the anode surface during charging, thus increasing energy density similarly to rechargeable batteries. The OPL_AC//NVPF_NF cell delivers a high specific energy of 326 Wh/kg at a specific power of 5729 W/kg. Even at an increased specific power of 115 kW/kg, the energy density remains at 31 Wh/kg (based on the total mass of active materials in both electrodes). Consequently, with their remarkable performance, RC-ASICs in a superconcentrated electrolyte system present a promising strategy for advancing energy storage devices.
