8. Moisture-Assisted Silylation Triggered by SiliconeAdhesives: A Hidden Electrolyte DecompositionMechanism in Supercapacitors.
Phatsawit Wuamprakhon, Nuttida Matkhaw, Thitiphum Sangsanit, Worapol Tejangkura, Santamon Luanwuthi, Pichamon Sirisinudomkit, Pawin Iamprasertkun, Jiaqian Qin, Montree Sawangphruk.
Journal : ChemSusChem (Chemistry Europe), 2026.
Impact Factor : 7.7, Q1, CiteScore : 15.8.
DOI: 10.1002/cssc.202501768.
Abstract : Silicone adhesives in polyimide (Kapton) tape are revealed as hidden initiators of electrolyte decomposition in commercial-scale supercapacitors employing acetonitrile-based electrolytes. This study uncovers a previously unrecognized, moisture-assisted silylation mechanism in which silicone-derived trimethylsilyl species react with acetamide, a hydrolysis product of acetonitrile, in the presence of triethylamine (TETA), forming trimethylsilyl acetamide (TMSA) via nucleophilic substitution. This degradation pathway, activated under elevated voltage (≥4.1 V) and trace moisture, is distinct from known electrode-induced processes and accelerates electrolyte breakdown. A suite of analytical techniques, including gas chromatography–mass spectrometry (GC–MS), X-ray fluorescence (XRF), X-ray photoelectron spectroscopy (XPS), and electrochemical testing, unambiguously identifies the silicone adhesive as the primary source of reactive silicon. Control experiments confirm that TMSA formation requires both silicone adhesives and water, validating the proposed mechanism. These findings challenge the conventional assumption that non-electroactive components are chemically inert and demonstrate that auxiliary materials can drive parasitic side reactions under realistic abuse conditions. This work highlights the critical importance of full-system material compatibility screening in supercapacitor design and provides mechanistic insight for enhancing device longevity and safety.
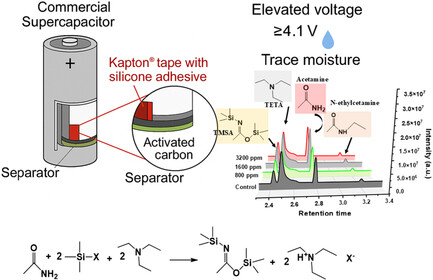
7. Solvation-Driven Interphase Engineering and Mechanical Failure Pathways in Large-Scale Anode-Free Lithium Metal Batteries.
Nattanon Joraleechanchai, Nuttida Matkhaw, Thitiphum Sangsanit, Worapol Tejangkura and Montree Sawangphruk.
Journal : Chemical Science, 2026, (Nature index).
Impact Factor : 7.4, Q1, CiteScore : 14.1, H-index 241.
DOI: 10.1039/D6SC00025H
Abstract : Anode-free lithium–metal batteries promise ultrahigh energy density but remain limited by unstable interfaces and safety concerns. Here, we demonstrate that fluorinated-ether solvation chemistry directly dictates interphase formation, thermal behavior, and performance in large-format Cu‖NMC90 cells. We reveal that introducing 1,1,2,2-tetrafluoroethyl 2,2,3,3-tetrafluoropropyl ether (TTE) into a localized high-concentration electrolyte restructures the Li⁺ solvation shell into a previously unreported PF₆⁻-dominated coordination environment in practical cylindrical cells. This anion-rich solvation promotes preferential PF₆⁻ reduction and yields a dense LiF-rich solid-electrolyte interphase, as verified by nuclear magnetic resonance, X-ray photoelectron spectroscopy, and differential electrochemical mass spectrometry. The optimized electrolyte enables non-flammable behavior and high efficiency in 18650-format anode-free cells, achieving 278–308 Wh kg⁻¹ at the cell level (380–402 Wh kg⁻¹ at the jelly-roll level), nearly 100% Coulombic efficiency, and compliance with UN38.3 impact-safety standards. Importantly, we identify lithium-plating-induced mechanical expansion—not interfacial instability—as the dominant failure pathway, establishing a direct link between molecular solvation structure and macroscopic structural integrity. These findings define solvation-driven interphase engineering as a practical route toward safe, high-energy anode-free lithium–metal batteries.

6. Fate of Tungsten-Coated NMC Cathodes in Li-Ion Cells.
Animesh Dutta, Kan Homlamai, Jeffin James Abraham, Thitiphum Sangsanit, Andrew O’Brien, Yixiang Zhang, Michel Johnson, Eytan Mendel-Elias, Montree Sawangphruk, JR Dahn.
Journal : ACS Energy Letters, 2026.
Impact Factor : 18.9, Q1, CiteScore : 29.6, H-index 223.
DOI: 10.1021/acsenergylett.6c00321
Abstract : To achieve high energy density in medium-nickel-layered oxide (NMC) cathodes, the most straightforward approach is to increase the upper cutoff voltage. However, this reduces the cycle life due to enhanced electrolyte oxidation. Many coating elements have been introduced to improve the lifetime, and tungsten (W) is a common vendor choice. We find that surface tungsten compounds dissolve during electrochemical cycling, with the rate increasing at higher cutoff voltages. X-ray photoelectron spectroscopy (XPS) confirms W deposition in the form of metallic W and tungsten oxides on the graphite negative electrode, and X-ray fluorescence (XRF) quantified W content. Surprisingly, a significant amount of W dissolves under high-voltage operation, a problem that becomes more severe because vendors generally employ only trace amounts of coating, which leaves the surface increasingly exposed as the coatings dissolve. This work also investigates the impact of deposited W on the lithiated graphite negative electrode through simulated storage experiments.

5. NaF-rich solid-electrolyte interphase formation stabilizes hard-carbon anodes in sodium-ion capacitors.
Phatsawit Wuamprakhon, Nuttida Matkhaw, Thitiphum Sangsanit, Worapol Tejangkura, Montree Sawangphruk
Journal : Chemical Engineering Journal, 2026.
Impact Factor : 13.2, Q1, CiteScore : 20.6, H-index 337.
DOI: 10.1016/j.cej.2026.173980
Abstract : Designing a stable and ionically conductive solid–electrolyte interphase (SEI) remains a central challenge for sodium-ion capacitors (SICs), where device performance is governed by interfacial transport kinetics rather than capacity retention alone. While fluorinated electrolyte additives are known to stabilize hard-carbon (HC) anodes in sodium-ion batteries, their kinetic consequences under the asymmetric architecture and high-rate operation of SICs remain poorly quantified. Here, we investigate the role of electrolyte additives in tailoring the SEI on HC anodes in SICs and establish a quantitative relationship between interphase chemistry, Na+ transport kinetics, and device-level performance. X-ray photoelectron spectroscopy depth profiling reveals that fluoroethylene carbonate (FEC) and a 1,3-propane sultone derivative (DTD) promote the formation of thin, NaF-rich inner SEI layers, whereas vinylene carbonate (VC), tris(trimethylsilyl) phosphite (TTE), and additive-free electrolytes yield thicker, organic-dominated interphases. Temperature-dependent electrochemical impedance spectroscopy and Arrhenius analysis demonstrate that FEC- and DTD-derived SEIs exhibit markedly reduced interfacial resistance growth and lower activation energies for Na+ transport compared to organic-rich SEIs. These kinetic advantages suppress parasitic electrolyte decomposition while minimizing polarization under capacitor-relevant operating conditions, leading to enhanced rate capability and long-term cycling stability. By correlating SEI composition, thickness, and Na+ transport activation barriers, this work clarifies why SEI chemistries that are chemically stable in sodium-ion batteries do not necessarily ensure optimal performance in SICs. Guided by these insights, optimized AC‖HC full cells with a 2:1 AC:HC mass ratio and pre-sodiated HC deliver ∼97.5% capacity retention after 400 cycles at 0.25 A g−1 with excellent rate performance. This study establishes kinetic-based design principles for SEI engineering in SICs and provides a framework for translating interfacial chemistry into performance-relevant metrics in high-rate sodium-based energy storage systems.

4. Enhancing The Performance of Medium Nickel Cathodes via Controlled Cobalt Rich Surface Coating.
Kan Homlamai, Animesh Dutta, Artem M Abakumov, Maria Kirsanova, Montree Sawangphruk, Yang Yu, Jeff R Dahn.
Journal : Journal of The Electrochemical Society, 2026, 173, 050509.
Impact Factor : 3.3, Q1, CiteScore : 6.1, H-index 321.
DOI : 10.1149/1945-7111/ae489a.
Abstract : Medium-nickel layered oxide (NMC) materials are widely adopted by cell manufacturers, as they provide higher energy density without compromising safety at high voltage. However, challenges such as poor rate capability and limited lifetime at higher cutoff voltages still hinder their broader application. Our analysis of vendor materials revealed that, among the multiple additives used to enhance performance, cobalt is also employed as a surface coating element by some vendors. This study demonstrates a simple process to achieve a cobalt-rich surface on the NMC cathode via an all-dry synthesis method, verified by X-ray Photoelectron Spectroscopy (XPS). Our Co-coated NMC samples achieve performance close to that of top-tier vendor samples that incorporate multiple coating additives. Transmission electron microscopy (TEM) further confirms the incorporation of cobalt on the surface of single-crystal Li1+x(Ni0.6Mn0.4)1-xO2. This work highlights the importance of a cobalt-rich surface, showing that minimal cobalt addition can significantly improve electrochemical performance at high cutoff voltages without requiring numerous additives.

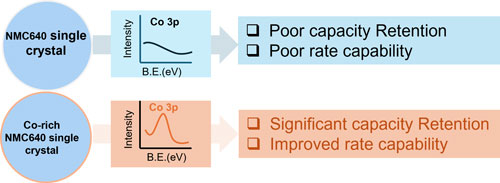
3. Interfacial and Structural Transformations in Ni-Rich Cathodes: A Roadmap Toward Chemical Stability.
Surasak Kaenket, Techin Mamiamuang, Nattanon Joraleechanchai, Jirawat Limphrasittisak, Purin Krapong, Worapol Tejangkura, Montree Sawangphruk.
Journal : Chemical Communications, 2026, (Nature index).
Impact Factor : 4.3, Q1 (87th), CiteScore : 10.2, H-index 387.
DOI : 10.1039/d5cc06328k.
Abstract : This review highlights Ni-rich layered oxide cathodes, such as LiNixMnγCozO2 (NMC) and LiNixCoyAlzO2 (NCA), where x ≥ 0.6, y + z ≤ 0.4, and x + y + z = 1, which have become the cornerstone of high-energy lithium-ion batteries due to their high specific capacities (>200 mA h g−1), reduced cobalt dependence, and compatibility with both cylindrical and pouch-cell formats. However, as Ni content exceeds 80%, these materials suffer from coupled chemical and mechanical degradation—cation disorder, oxygen loss, and interfacial instability—that limits lifetime and safety. This feature article presents a comprehensive roadmap linking the mechanistic origins of degradation to scalable mitigation strategies, bridging fundamental insights and technology readiness level (TRL) 9 implementation. At the lattice level, antisite defects (Ni2+/Li+ mixing) and anisotropic H2–H3 phase transitions generate microstrain and intergranular cracking, which are effectively mitigated through bulk doping (e.g., W6+, Ti4+, Zr4+, Sc3+), co-doping, and single-crystal or columnar morphologies that distribute internal stress. At the electronic level, excessive delithiation triggers oxygen redox and lattice-oxygen release, initiating chemomechanical collapse and surface rock-salt reconstruction. Countermeasures include oxygen-constraining coatings, Li2NiO2 prelithiation, and redox-buffering additives (e.g., LiFePO4 blending). At the interface, parasitic reactions with carbonate electrolytes produce resistive cathode–electrolyte interphases (CEIs) and gas evolution. Stabilization is achieved via fluorine-rich electrolytes, hybrid compartmentalized systems, and MOF-functionalized separators, which suppress HF formation and transition-metal dissolution. The article further highlights emerging manufacturing-compatible solutions—including solvent-free mechanofusion coatings, spatial atomic layer deposition, facing-target sputtering, and wet-chemical nanoshell growth—that integrate surface and bulk stabilization. These approaches not only improve high-voltage cycling (>4.5 V) but also meet industrial scalability and sustainability goals through direct regeneration and closed-loop cathode recycling. By unifying lattice, oxygen, and interfacial stabilization into a coherent framework, this roadmap provides actionable guidance for designing next-generation Ni-rich cathodes that achieve long-term durability, high safety, and industrial manufacturability for the global electrification era.

2. Graphene in energy harvesting devices.
Montree Sawangphruk.
Graphene: Synthesis, Properties, Technology and Applications, 2026, 157-183.
Abstract : This chapter provides a comprehensive role of graphene in energy-harvesting devices, covering its integration into various technologies and applications. Starting with the introduction of graphene and its exceptional properties, this book delves into its potential in enhancing photovoltaic cells by improving efficiency and performance through the use of transparent graphene electrodes as well as in heterojunction and organic solar cells. The book then examined the role of graphene in supercapacitors, highlighting its contribution to increased energy density, faster charge/discharge rates, and enhanced durability. Employing graphene in lithium-ion batteries is also discussed, emphasizing its potential to improve the capacity, charge times, and overall performance through innovative anode and cathode materials. Thermoelectric devices benefit from graphene’s superior thermal and electrical conductivities, with applications in waste heat recovery and power generation. Graphene-enhanced piezoelectric and triboelectric devices have been explored, demonstrating their ability to efficiently harvest mechanical energy. Real-world applications and case studies illustrate the practical impact of graphene-based energy solutions from wearable electronics to renewable energy systems. The book addresses the challenges of large-scale integration, material compatibility, and environmental sustainability and outlines future research directions focusing on material innovation and scalable production techniques. Finally, the book highlights the potential global impact of graphene-based energy-harvesting devices on energy efficiency, renewable energy integration, and green society.
1. Decoupling Oxygen Redox from O₂ Release in Li-and Mn-Rich Layered Cathodes: Mechanisms, Metrics, and Design Rules.
Techin Mamiamuang and Montree Sawangphruk.
Journal : Journal of Materials Chemistry A, 2026.
Impact Factor : 9.5, Q1, CiteScore : 16.7, H-index 318.
DOI : 10.1039/D5TA07671D.
Abstract : Lithium- and manganese-rich (LMR) layered oxides can deliver >250 mAh g⁻¹ by engaging anionic (oxygen) redox, yet their promise is undermined when oxygen redox couples to O₂ formation, triggering transition-metal migration, layered→spinel/rock-salt reconstruction, interfacial breakdown, and voltage fade. This review reframes LMR development around a single objective—decouple reversible oxygen redox from O₂ release—and organizes the field into mechanisms, metrics, and design rules. We first clarify the mechanistic pathways that produce oxidized-oxygen species versus molecular O₂ and map how these pathways propagate stress, porosity/voids, and interfacial reactivity. We then define a decision-grade metric set to distinguish O-redox from O₂ evolution under practical conditions, including gas quantification at realistic cutoffs (≥4.5 V), operando O-species fingerprints (e.g., RIXS/¹⁷O probes), proxies for transition-metal migration, and tracking of microstructural change (voids, reconstruction, impedance growth). Finally, we translate diagnostics into actionable design rules spanning (i) bulk/composition (Mn-valence control, Li/TM ordering, concentration gradients, high-entropy chemistries), (ii) architecture and interfaces (primary-particle coatings; thin, Li⁺-conductive, acid-scavenging layers; oxygen-tolerant CEIs), and (iii) electrolytes (fluorinated and localized-high-concentration systems with targeted additives). Emerging concepts—dynamic oxygen buffers, self-regenerating interphases, and solid/gel interlayers—are assessed against application-relevant benchmarks (areal loading, temperature, gas evolution, N/P balancing, scalable synthesis). We conclude with prioritized experiments and go/no-go criteria to accelerate durable, high-voltage LMR commercialization.

